Scientists are continuously working on to find an alternative to fossil fuels. We are familiar with the side effects of fossil fuels and its impact on environment. Right now the fossil fuels seem to be necessary evil we can’t do without. Scientists want alternative fuels that have none of the ill effects of traditional fuels.
Scientists are continuously working on to find an alternative to fossil fuels. We are familiar with the side effects of fossil fuels and its impact on environment. Right now the fossil fuels seem to be necessary evil we can’t do without. Scientists want alternative fuels that have none of the ill effects of traditional fuels. For a long time researchers are eying the carbon dioxide as an alternative fuel. Because carbon dioxide emanation is a hotly-debated global issue. This gas is held responsible for greenhouse effect that in turn is causing irreversible climate change. Human beings are responsible for carbon dioxide emissions. This gas is produced when we use fossil fuels. This is not the exact figure but carbon dioxide’s amount is increasing in earth’s atmosphere by more than 30% since the industrial revolution. According to estimates of The Intergovernmental Panel on Climate Change (IPCC) the world requires to reduce the global carbon dioxide by 60% in order to stabilize it at present-day levels.
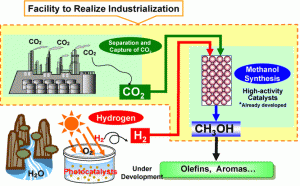
Scientific researches are focusing on solutions that reduce global warming. Presently there is one popular term known as “green chemistry” which aims at minimizing or altogether eliminating the use of hazardous substances to prevent environmental pollution. Scientists at the Singapore-based Institute of Bioengineering and Nanotechnology (IBN) are striving to make the mass production of methanol more cost-effective. This will result in reducing the amount of carbon dioxide released in the earth’s atmosphere. Scientists at the Singapore-based Institute of Bioengineering and Nanotechnology (IBN) have achieved an unparalleled feat by transforming carbon dioxide into methanol. Methanol is a widely used form of industrial feedstock and clean-burning biofuel.
Their main achievement was using “organocatalysts” and making the whole process non-toxic to produce the more useful chemical compound. Professor Jackie Y. Ying who is IBN executive director of the Institute of Bioengineering and Nanotechnology shares his views, “We are innovating effective methods of generating clean energy using green chemistry and nanotechnology. In the face of environmental pollution, global warming and increasing demands on diminishing fossil fuel resources, we hope to provide a viable alternative energy option for industry, and effective sequestration and conversion of carbon dioxide.”
At the IBN, scientists successfully made carbon dioxide react with a stable organocatalyst called N-heterocyclic carbene (NHC). This reaction took place under mild conditions in dry air. Siti Nurhanna Riduan who is the senior lab officer at IBN explained about the experiment, “NHCs have shown tremendous potential for activating and fixing carbon dioxide. Our work can contribute towards transforming excess carbon dioxide in the environment into useful products, such as methanol.”
Further they used a combination of silica and hydrogen known as hydrosilane. Hydrosilane is added to the NHC-activated carbon dioxide, which is converted into methanol through hydrolysis. Dr Yugen Zhang who is the IBN team leader and principal research scientist elaborates about the intricacies of the chemical reaction, “Hydrosilane provides hydrogen, which bonds with carbon dioxide in a reduction reaction. This carbon dioxide reduction is efficiently catalyzed by NHCs even at room temperature. Methanol can be easily obtained from the product of the carbon dioxide reaction.”